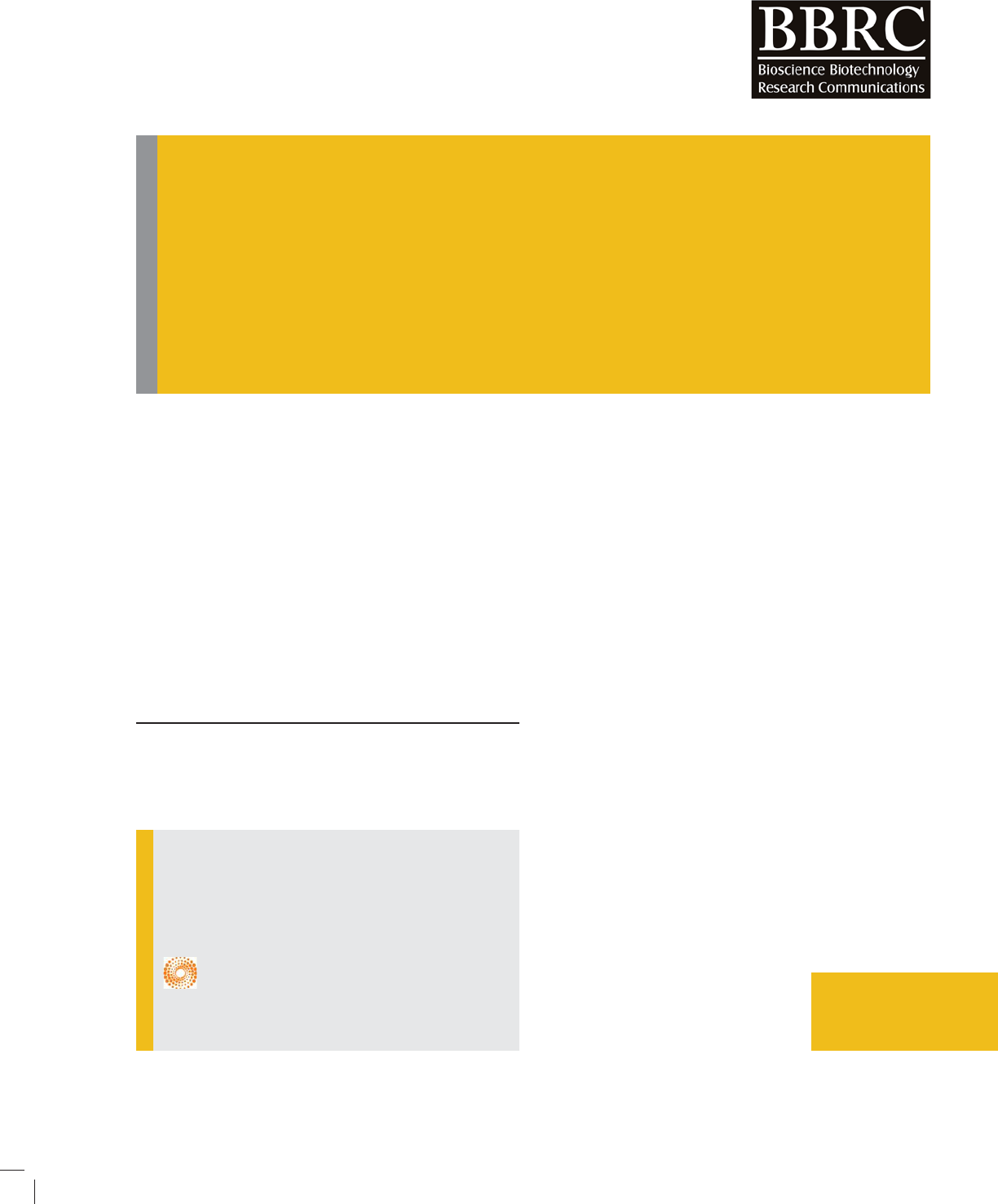
Biotechnological
Communication
Biosci. Biotech. Res. Comm. 9(3): 489-494 (2016)
Mathematical modeling of microbial growth and
production kinetics for -amylase production using
mustard oil cake as solid substrate
Santosh K. Mishra
a
, Shashi Kumar
b
, Ravi Kant Singh
c
* and Surendra Kumar
b
a
Department of Biotechnology, IMS Engineering College, Ghaziabad, UP, India
b
Chemical Engineering Department, Indian Institute of Technology Roorkee, Roorkee 247667, Uttarakhand,
India
c
*Department of Biotechnology, Noida Institute of Engineering & Technology, Greater Noida, UP, India
ABSTRACT
Different phases of the Gliomastix indicus growth curve and the production of -amylase using solid-state fermen-
tation process based on variation in dry weight was mathematically modeled. The result of the study reveals that
the growth of the fungal cells and the production of -amylase on a mustard oil cake as solid substrate could be
expressed by simple models incorporating the mathematical de nition of each phase and the variation in dry sub-
strate weight over the incubation time. The growth kinetics of G.indicus could be described by the mathematical
modeling parameters regarding maximum speci c growth rate and maximum biomass concentration obtained by
tting the experimental data to the logistic model. Experimental data collected from a series of batch fermentations
process were collected for 10 days (240 hrs.) and used to propose the mathematical models. Experimental observa-
tions, and predicted models made it possible to conclude that these models can be successfully employed to represent
the biomass growth and -amylase production in solid-state fermentation processes.
KEY WORDS: SOLID STATE FERMENTATION, MATHEMATICAL MODELING, BIOMASS GROWTH KINETICS, PRODUCTION KINETICS
489
ARTICLE INFORMATION:
*Corresponding Author: rksingh.iitr@hotmail.com
Received 25
th
July, 2016
Accepted after revision 5
th
Sep, 2016
BBRC Print ISSN: 0974-6455
Online ISSN: 2321-4007
Thomson Reuters ISI ESC and Crossref Indexed Journal
NAAS Journal Score 2015: 3.48 Cosmos IF : 4.006
© A Society of Science and Nature Publication, 2016. All rights
reserved.
Online Contents Available at: http//www.bbrc.in/
INTRODUCTION
Solid state fermentation process (SSF) involves the
growth of microorganisms on a moist insoluble solid
substrate in the absence or sometimes nearly absence of
free liquid (Mitchell et al 2002; Pandey 2003). The level
of water available for growth of microorganism in SSF
is low which makes it suitable for the growth and culti-
vation of fungi (Pandey 2003; Cannel and Moo Young
1980). The solid state fermentation (SSF) is used in a
number of fermentation industries for the production of
enzymes, organic acids, and other bioactive compounds.

490 MATHEMATICAL MODELING OF GROWTH AND PRODUCTION OF -AMYLASE BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS
Santosh, Shashi, Ravi Kant
Fermentation processes can be improved by using suit-
able methods to estimate the biomass and other main
process variables resulting in the investigation of the
associations between growth kinetics and the fermen-
tation product. In SSF precise estimate is complicated
because of the dif culty in the separation of biomass
from the fermentation media. Therefore, appropriate
data are not accessible concerning the kinetics and bio-
mass in SSF.
Substrate moisture content, temperature and, bio-
mass have been found as the critical variables that
affect growth of microorganism and enzyme production
(Khanahmadia et al.2006; Desgranges et al.1991) For
any SSF these parameters must be controlled during the
entire fermentation processes. Several approaches have
been proposed by researchers for indirect measurements
of biomass formation, such as estimating the production
of primary metabolic product (Desgranges et al.1991;
Okazaki et al.1980) amount of carbon dioxide produc-
tion (Raimbault 1998), the variation in the electrical
conductivity of the biomass and solid substrate (Carri-
azalez et al.1981) and the changes observed in the color
of the fermentation medium. Other than these methods,
a direct method of viable cell count can also be used to
estimate the amount of biomass formation.
There are consequential dif culties that have been
observed with the direct measurement of cell biomass in
SSF systems therefore a weighing method could provide
a signi cant method in kinetic studies of such processes.
Bioprocess modeling including cells and cultures can
be of signi cant importance in optimizing and control-
ling actual production process of biomass and product
(Curien et al. 2003; Grosz and Stephanopoulos 1999).
Several challenges have been observed at each stage in
the development of models of enzyme production kinet-
ics (Copella and Dhurjati 1989; Thilakavathi et al. 2007).
Earlier, a kinetic model has been developed for batch
fermentation for lactic acid production using cane-
sugar molasses by Enterococcus faecalis. Parameters of
the kinetic model have been determined and validated
based on experimental data by using genetic algorithm
(Nandsana and Kumar 2008, Gelatin et al. 2015).
The effect of temperature and substrate moisture
content on the growth and production of amylase, pro-
tease and phytase by Aspergillus niger during SSF was
investigated and a mathematical model for the kinet-
ics of growth and enzyme production were developed
earlier. A mathematical model to describe the kinetics
of enzyme production by the lamentous fungal sp.
Trichoderma harzianum was developed using sugarcane
bagasse as solid substrate for the production of cel-
lulase, beta glucosidase and xylanase. In all the stud-
ies, it has been found that temperature and substrate
moisture content of the media play a signi cant role for
the growth of microorganism and enzyme production
(Saithi et al. 2016).
In recent years, SSF has gained renewed interest
from researchers for the production of enzymes which
have industrial important in view of its economic and
engineering advantages. In this work we are propos-
ing a mathematical model can be used as an aid for
improving the design and control of SSF processes. The
major objective of this study was to identify and develop
model for the growth, stationary, and death phases of the
growth curve and -amylase production kinetics dur-
ing SSF processes using a comparatively simple weigh-
ing method by using G. indicus as a fungal strain. The
knowledge gained from our research work may contrib-
ute to the understanding and control of SSF processes
for large scale enzyme production using fungal strain.
MATERIAL AND METHODS
Inoculum preparation: The microorganism Gliomastix
indicus (MTCC 3869) was procured from the Institute of
Microbial Technology (IMTECH) Chandigarh India. G.
indicus comprises the properties of lamentous fungi
and rapidly grows on all common mycological media
such as malt extract, potato dextrose agar etc. This fun-
gal strain was maintained on the potato dextrose agar
(PDA) medium which contains; potatoes 200g/l, dex-
trose 20g/l and Agar 15g/l. The pH of the medium was
adjusted to 6 by using 1N NaOH. The organism was
maintained by the serial transfer on the PDA medium
after every fortnight and incubated at 30°C.
Inoculum was prepared by transferring 5ml suspen-
sion culture, into 250ml conical ask containing 95ml
of sterile inoculum medium. The composition of the
inoculum medium was (g/l): Glucose (20g/l), NH
4
NO
3
(3g/l), MgSO
4
.7H
2
O (0.5g/l), KCl (0.5g/l), K2HPO
4
(1g/l),
FeSO
4
.7H
2
O (0.01g/l), with a pH of solution 6.0. The
asks were incubated on a rotary shaker at 200rpm at
28°C for 48 hrs (Nagalaxmi et al. 2008; Kammoun et al
2008).
Cultivation of Fungal Strain: G.indicus was cultivated
in SSF using mustard oil cake as solid substrate and
moistened with distilled water (1:1.2). After autoclaving
at 121˚C for 20 min, the culture medium was inoculated
with 15% (v/v) of the above applied inocula and mixed
thoroughly to ensure uniform distribution. The inocu-
lated MOC was distributed in Petri dishes (8 cm in diam-
eter) as follows: 1.0 cm average thickness of the culture
medium, 10 g initial weight of inoculated medium per
dish, 80% initial moisture content, and incubated at
30˚C for 0-240 hr. Dish samples were removed from the
incubator at regular intervals for the determination of
the number of viable cells, the total dry weight of the
fermented substrate, and the production of -amylase.

BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS MATHEMATICAL MODELING OF GROWTH AND PRODUCTION OF -AMYLASE 491
Santosh, Shashi, Ravi Kant
Analytical methods: Viable cell count: The viable
bacterial count was determined by suspending the fer-
mented substrate in peptone-phosphate buffer at a 1:10
ratio and shaking the suspension for 15 min, followed
by serial dilution and plating onto plate count agar. The
number of viable colonies in the fermented substrate
after 96 h incubation at 30°C is reported as cfu/g.
Total dry weight: The total dry solid weight per dish
was evaluated by drying at 80 ± 3°C to constant weight.
Then the nal dry weight was calculated and presented
as the variation in the dry matter of the fermented wheat
bran during the test as Wt.
-amylase activity: Crude enzyme was extracted by
mixing a known quantity of fermented substrate with
distilled water in a 1:10 ratio on a rotary shaker (180
rpm) for 1 h. The slurry was squeezed through wet
cheesecloth. Then the ltrate was centrifuged (10,000 ×
g, 10 min, 4 °C), and the clear supernatant was used as
the source of the crude enzyme. The activity of -amylase
was determined by the Bernfeld procedure using soluble
starch (Qualigens) as a substrate (Bernfeld 1955). The
reaction mixture was incubated for 15 min at 35˚C. One
unit (U) of -amylase is de ned as the amount of enzyme
that releases 1 mol of reducing sugar as maltose per
minute under the assay conditions and is expressed as
U/g of dry fermented substrate.
Mathematical Model:The total weight of medium
under SSF using MOC as solid substrate can be written
as:
Total Wt (W) = Wt of Solid substrate(S)
+Wt of Biomass(B)+ Wt of Product(P) …(1)
The value of all the three parameters will change as the
fermentation process progress. Hence above equation
can be represented in differential form with respect to
time. Therefore:
…(2)
The substrate consumption rate by biomass will be dif-
ferent in different growth stages. Hence rate of substrate
consumption with respect to time under different stages
can be represented as:
…(3)
The substrate consumption rate under different growth
phase can be de ned by either biomass growth equation
and biomass yield coef cient or by using enzyme pro-
duction equation and coef cient of yield (Amrane and
Pregent 1994; Venkatesh et al 1993; Yeah et al. 1991)
As suggested by Hashemi et.al, the equation for the
estimation of total biomass weight with respect to total
medium weight can be represented as:
…(4)
Furthermore, the equation for the estimation of total
weight of product can also be represented as:
…(5)
The above equations indicate that the amount of
biomass and product concentration in the fermentation
media can be estimated with the help of total value of W
(Hashemi et al 2011).
RESULT AND DISCUSSION
Change in dry weight of medium during the fermenta-
tion process was calculated by substracting the weight
recorded each day for continuously ten days from the
initial weight i.e. W
0
=10.3 gm as shown in gure-1.
Growth phase was observed during the time period
of 0-96 hr, stationary phase was found to be between
96-144 hr and subsequently decline phase was observed
between 144-240 hr onwards. Now the values of growth
phase and decline phase time frames were used to get
the following equations respectively:
…(6)
…(7)
For the above Eqs. (6) and (7) coef cients of determinant
(R
2
) were found to be 0.842 and 0.979. Along with it,
the residual sums of squares (rss) for both the equations
were 0.302 and 0.133 respectively. These values indi-
cates that the Eqs. correlate the experimental data with
a very approximation.
We can differentiate the Eqs. (6) and (7) with respect
to time in order to get the relation for time dependent
total medium weight degradation.
…(8)
…(9)
492 MATHEMATICAL MODELING OF GROWTH AND PRODUCTION OF -AMYLASE BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS
Santosh, Shashi, Ravi Kant
FIGURE 1. Change in dry weight of medium observed during
fermentation
Eqs. (4) and (8) represent the equation for time depend-
ent medium weight variation during biomass growth
phase. Similarly, Eqs. (4) and (9) represent the equation
for time dependent medium weight variation during bio-
mass death phase. If the values of and are known,
Eqs. (4) and (8) can be used for the calculation of bio-
mass dry weight (B) during growth phase. Similarly, Eqs.
(4) and (9) can also be used for the calculation of B dur-
ing death phase.
The and values depend upon the selection of
medium, choice of microorganism and the environmental
conditions of fermentation process and it can be experi-
mentally determined. For representation, the trend of
growth curve based on the derived mathematical equa-
tion, / value was assumed to be 0.01. Initial biomass
dry weight (B
0
)was experimentally determined and found
to be 0.0042 g. Hence, Eqs. (8) and (4) will give:
…(10)
Therefore, the nal Eq. for biomass growth in growth
phase will be:
…(11)
As per the earlier assumptions t=96-144 were considered
as stationary phase. Hence, B
144
= B
96
= 0.0052 g. Eqs. (9)
and (4) will give:
…(12)
Therefore, the nal Eq. for biomass growth in death
phase will be:
…(13)
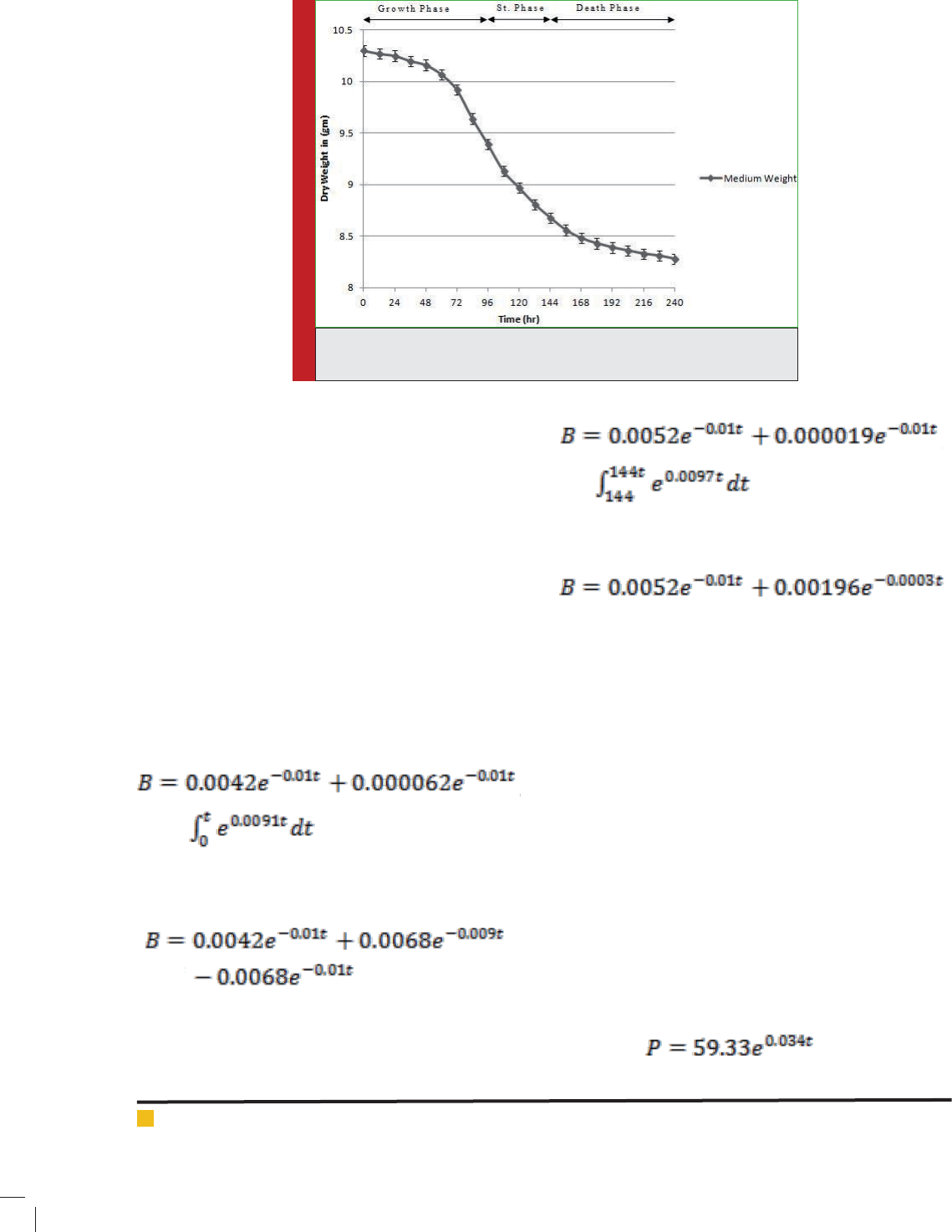
Using equation (11) and (13) and considering the time
period of growth, stationary and death phase, the calcu-
lated values were plotted as shown in gure-2.
As shown in the gure, the calculated values on the
basis of medium dry weight variation are in good cor-
relation with the experimental values.
The mathematical model for -amylase production
kinetics in SSF is very convenient to represent the com-
plex problem of such process. Here we are introducing
a model for the prediction of -amylase production
kinetics on solid substrate using fungal strain G. indi-
cus during fermentation process. Eq. (5) correlates the
total product (P) with respect to fermentation time (t)
using weight variation data of medium. The total prod-
uct i.e. -amylase was assayed for the entire period of
fermentation process at regular time intervals. Follow-
ing Eqs. (14), (15) and (16) were derived to represent the
-amylase production during growth phase, stationary
phase and death phase respectively:
…(14)
BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS MATHEMATICAL MODELING OF GROWTH AND PRODUCTION OF -AMYLASE 493
Santosh, Shashi, Ravi Kant
…(15)
…(16)
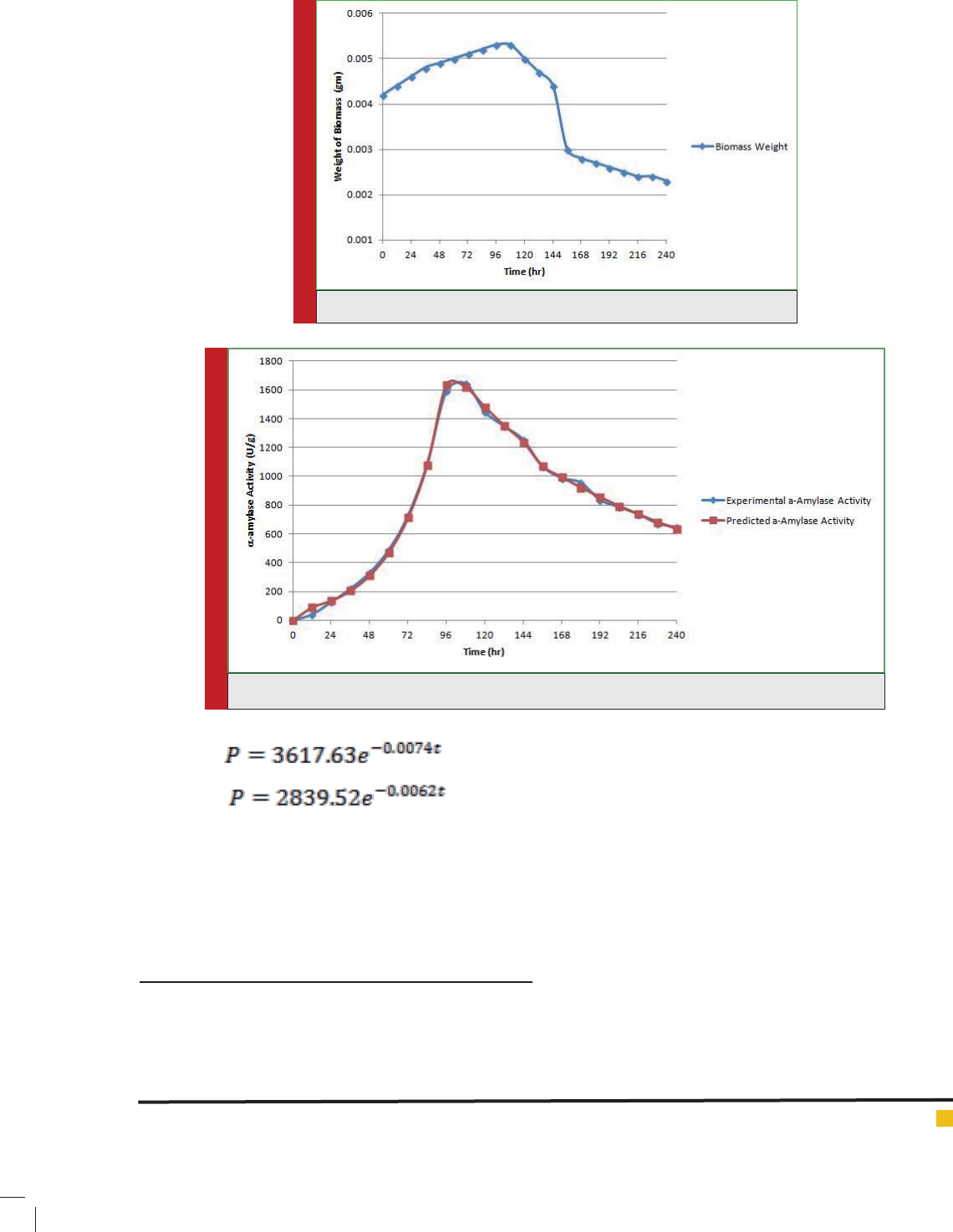
Form the above Eqs. (14), (15) and (16), the coef cients of
determination (R2) were calculated as 0.99, 0.97 and 0.98
respectively. Figure-3 represents the correlation between
experimental and predicted data calculated from the
product kinetic mathematical model given above.
CONCLUSION
Because of the complexity of SSF systems, estimation of
the biomass and product formation during the incuba-
tion time could give highly valuable information leading
to a more comprehensive understanding of such compli-
cated systems. We investigated a feasible approach for
modeling the different phases of the fungal growth curve
and production of -amylase as product by G. indicus
in a SSF process based on variation in dry weight. The
result showed that fungal growth and the production of
-amylase on a MOC as solid substrate could successfully
be modeled based on variations in solid substrate weight
and can be used for large scale industrial production.
REFERENCES
Amrane A, Prigent Y (1994) Mathematical model for lactic acid
production from lactose in batch culture: model development
and simulation. J. Chem. Technol. Biotechnol. 60:241–246
Bernfeld P (1955) Amylase and Methods Enzymol. 1:149–158
Cannel E, Moo-Young (1980) Solid-state fermentation systems.
Process. Biochem. 4:2-7
FIGURE 2. Change in dry weight of biomass with time variation
FIGURE 3. Predicted and experimental -amylase activity

Santosh, Shashi, Ravi Kant
494 MATHEMATICAL MODELING OF GROWTH AND PRODUCTION OF -AMYLASE BIOSCIENCE BIOTECHNOLOGY RESEARCH COMMUNICATIONS
Carrizalez V, Rodríguez H, Sardina I (1981) Determination of
the speci c growth of molds on semi-solid cultures. Biotech-
nol. Bioeng. 23:321–333
Coppella SJ, Dhurjati P (1989) A detailed analysis of Saccha-
romyces cerevisiae growth kinetics in batch, fed-batch, and
hollow- ber bioreactors. Chem Eng. J. 41:B27–B35
CurienG, Ravanel S, Dumas R (2003) A kinetic model of the
branch-point between the methionine and threonine biosyn-
thesis pathways in Arabidopsis thaliana. Eur. J. Biochem. 270
:4615–4627
Desgranges C, Georges M, Vergoignan C,Durand A (1991) Bio-
mass estimation in solid state fermentation. II. On-line meas-
urements, Appl. Microbiol. Biotechnol. 35:206–209
Desgranges C, Vergoignan C, Georges M, Durand A(1991)
Biomass estimation in solid state fermentation I. Manual
biochemical methods, Appl. Microbiol. Biotechnol. 35:200–
205
Gelain L, da Cruz Pradella JG, da Costa AC (2015) Mathemati-
cal modeling of enzyme production using Trichoderma harzi-
anum P49P11 and sugarcane bagasse as carbon source.Biore-
source technology.198:101-107
Grosz R, Stephanopoulos G (1999) Physiological, biochemical,
and mathematical studies of micro-aerobic continuous ethanol
fermentation by Saccharomyces cerevisiae. III: mathematical
model of cellular energetics and catabolism. Biotechnol. Bio-
eng. 36:1030–1040
Hashemi M, Mousavi SM, Razavi SH ,Shojaosadati SA(2011)
Mathematical modeling of biomass and
-amylase produc-
tion kinetics by Bacillus sp in solid-state fermentation based
on solid dry weight variation. Biochemical Engineering Jour-
nal,53:159-164
Kammoun R, Naili B, Bejar S (2008) Application of a statistical
design to the optimization of parameters and culture medium
for
-amylase production by Aspergillus oryzae CBS 819.72
grown on gruel (wheat grinding by-product). Bioresource
Technology 99:5602-5609
Khanahmadia M, Roostaazad R,Mitchell DA, Miranzadeh M,
Bozorgmehri R, Safekordi A (2006) Bed moisture estimation by
monitoring of air stream temperature rise in packed-bed solid-
state fermentation. Chem. Eng. Sci. 61:5654–5663
Mitchell DA, Berovic M, Krieger N(2002) Overview of solid
state bioprocessing. Biotechnol Annu. Rev. 8:183–225
Nagalaxmi S, Vijayalakshmi M, Subrahmanyam A (2009) Glio-
mastix indicus sp nov. Current Trends in Biotechnology and
Pharmacy 3:111-112
Nandasana AD, Kumar S (2008) Kinetic modeling of lactic acid
production from molasses using Enterococcus faecalis RKY1.
Biochemical Engineering Journal. 38:277–284
Okazaki N, Sugama S, Tanaka T (1980) Mathematical model of
surface culture of koji mould. J Ferment. Technol. 51:471–476
Pandey A (2003) Solid-state fermentation. Biochem. Eng. J.
13:81–84
Raimbault M (1998) General and microbiological aspects of
solid substrate fermentation.Electronic Journal of Biotechnol-
ogy 3:26-27
Saithi S, Borg J, Nopharatana M, Tongta A (2016) Mathemati-
cal Modeling of Biomass and Enzyme Production Kinetics by
Aspergillus niger in Solid-State Fermentation at Various Tem-
peratures and Moisture Contents. J. Microb. Biochem. Technol.
8: 123-130
Thilakavathi M, Basak T, Panda T(2007) Modeling of enzyme
production kinetics. Appl. Microbiol. Biotechnol. 73:991–1007
Venkatesh KV, Okos MR, Wankat PC (1993)Kinetic model of
growth and lactic acid production from lactose by Lactobacil-
lus bulgaricus. Process Biochem. 28:231–241
Yeh PLH, Bajpai RK, Iannotti EL (1991) An improved kinetic
model for lactic acid fermentation, J. Ferment. Bioeng. 71: 75–77